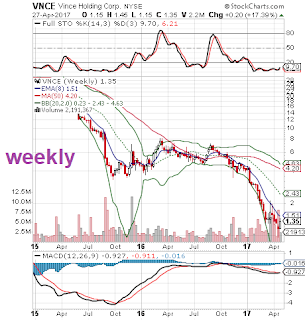
** charts before earnings **
** charts after earnings **
Vince Holding misses by $0.10, misses on revs; discloses going concern, material weakness; suspends guidance :
- Reports Q4 (Jan) loss of $0.15 per share, excluding $3.13/share impairment, $0.10 worse than the two analyst estimate of ($0.05); revenues fell 21.9% year/year to $63.9 mln vs the $74.55 mln two analyst estimate.
- Wholesale segment net sales decreased 28.4% to $34.4 million primarily due to a reduction in both off-price and replenishment orders, as well as an increase in allowances. Direct-to-consumer segment net sales decreased 12.6% to $29.4 million compared to the fourth quarter of fiscal 2015. Comparable sales decreased 20.5%, including e-commerce sales, as a result of declines in the number of transactions, due to reduced traffic, and a decrease in average order value.
- In accordance with new accounting guidance that became effective for the Company's fiscal year ended January 28, 2017, management has concluded that there is substantial doubt about the Company's ability to continue as a going concern for the twelve months following the date that the financial statements are issued, specifically relating to its ability to comply with the consolidated net total leverage ratio under its term loan facility. The Company's assessment did not take into account management's plans to mitigate such substantial doubt that could be reasonably possible of occurring but are not final, including discussions with lenders and with its majority shareholder on additional financing options and actions to improve the capital structure of the Company. As of January 28, 2017, the Company was in compliance with applicable financial covenants.
- As a result of management's assessment of the effectiveness of the Company's internal control over financial reporting as of January 28, 2017, the Company identified deficiencies in internal control over financial reporting that were assessed as material weaknesses. The Company has subsequently performed additional analysis, substantive testing and other post-closing procedures to ensure that its consolidated financial statements were prepared in accordance with U.S. GAAP. Accordingly, management believes that its consolidated financial statements fairly present its financial condition, results of operations and cash flows for the periods stated, in all material respects. The Company is taking specific steps to remediate these material weaknesses by implementing and enhancing its control procedures. These material weaknesses will not be remediated until all necessary internal controls have been implemented, tested and determined to be operating effectively.
- "As we look to 2017, we have made the prudent decision to suspend our sales and EPS guidance as we work to make our new systems more efficient and complete our business transition. This decision to suspend guidance was further driven by the difficult retail environment in which we continue to operate. That said, we remain focused on expanding our direct-to-consumer business, optimizing our wholesale channel, and growing our international presence over the long-term."